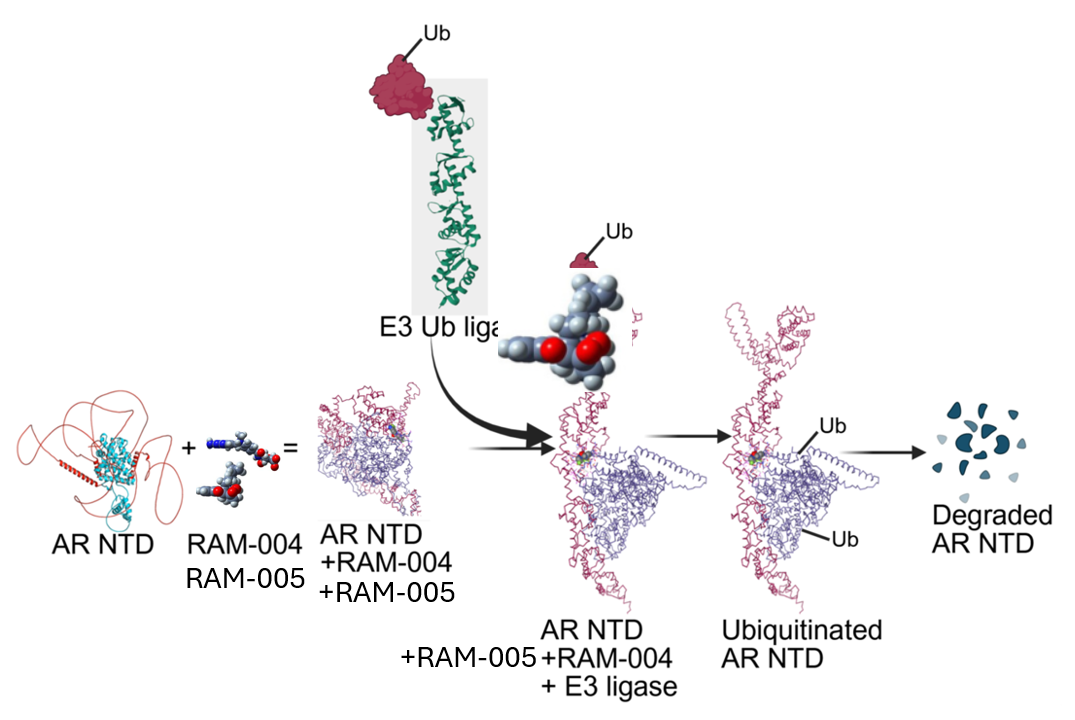
Molecular glues degraders are small molecules that induce or stabilize interactions between a target protein and an E3 ligase, leading to selective degradation of disease-causing proteins. Unlike traditional inhibitors, they degrade the disease-causing proteins by leveraging the ubiquitin, proteasome system.